What Phase Is Covid 19 Vaccine In Texas
The core argument from Hall and other vaccine skeptics is that the COVID-19 shot is a. ImmunityBio launched a Phase 1 trial of a Covid-19 vaccine in October 2020 in the United States and another in South Africa in January.

Collin County To Reopen Vaccine Sign Ups On Friday Wfaa Com
NVAX today announced that NVX-CoV2373 its recombinant nanoparticle protein-based COVID-19 vaccine demonstrated 100 protection against moderate and severe disease 904 efficacy overall and met the primary endpoint in its PREVENT-19 pivotal Phase 3 trial.

What phase is covid 19 vaccine in texas. COVID-19 vaccine protects monkeys from new coronavirus Chinese biotech reports. Everyone age 12 and older is now eligible to receive a COVID-19 vaccine in Texas. By Jon Cohen Apr.
A COVID19 vaccine is a vaccine intended to provide acquired immunity against severe acute respiratory syndrome coronavirus 2 SARSCoV2 the virus that causes coronavirus disease 2019 Prior to the COVID19 pandemic an established body of knowledge existed about the structure and function of coronaviruses causing diseases like severe acute respiratory syndrome SARS and Middle. Texas town doubled its COVID-19 vaccination rate in a. Who can sign up for a COVID-19 vaccine in Texas Phase 1C.
Phase 1C gives people who are at. GAITHERSBURG Md June 14 2021 Novavax Inc. For Vaccine Development at Texas Childrens Hospital and serves as.
Why vaccine passports may be inevitable in next phase of COVID-19. Texas Phase COVID-19 Vaccine Priorities. The study is designed to primarily evaluate the efficacy safety and immunogenicity of mRNA-1273 to prevent COVID-19 for up to 2 years after the second dose of mRNA-1273.
Animal trials and the first phase of. Food and Drug Administration in December 2020 most. All vaccines are authorized for people age 18 and older.
Phase 1 Trial of an RNA-based Covid-19 Vaccine Two doses 3 weeks apart of a lipid nanoparticle nucleoside-modified RNA vaccine encoding a trimerized SARS-CoV-2 receptorbinding domain elicited. In March 2020 the World Health Organization WHO declared coronavirus disease 2019 COVID-19 which is caused by severe acute respiratory syndrome coronavirus 2 SARS-CoV-2 1 a pandemicWith rapidly accumulating numbers of cases and deaths reported globally 2 a vaccine is urgently neededHere we report the available safety tolerability and immunogenicity data from an ongoing. If you continue to receive this error please contact your Tableau Server Administrator.
W hen the first COVID-19 vaccines from Pfizer-BioNTech and Moderna were authorized by the US. As of Wednesday May 12 2021 everyone ages 12 and older is now eligible to receive a COVID-19 vaccine in Texas. An unexpected error occurred.
The mRNA-1273 vaccine is being developed to prevent COVID-19 the disease resulting from Severe Acute Respiratory Syndrome coronavirus SARS-CoV-2 infection. All adults 50 KXAN March 15 2021. The Pfizer and Moderna vaccines are authorized for people 12 and older.
Texas equally will strive to ensure vaccine reaches communities with health disparities in accordance with Texas Vaccine Allocation Guiding Principles. 23 2020 105 PM. Vaccine so they will be protected from COVID- 19 while on the job.
Phase 1A4 Additional Outpatient Lab Specimen Testing and Reporting COVID-19 Research Additional Outpatient Faculty staff house staff and students providing direct patient care in an outpatient non-procedural location who cannot work from home and were not vaccinated in. The Texas Department of State Health Services expanded the vaccine allocation to a new group of people Monday. Their resistance to getting a COVID-19 vaccine.
A phase 123 placebo-controlled randomized observer-blind dose-finding study to evaluate the safety tolerability immunogenicity and efficacy of sars-cov-2 rna vaccine candidates against covid-19 in healthy individuals. In February 2021 the company registered a Phase. Communication and outreach will encourage vaccine uptake among these populations during Phase 1B.
The current vaccines even those with EUAs continue through a trial phase where they are tracking their volunteers to learn more about the long-term outcomes of taking the vaccine. European drugmakers Sanofi and GSK kicked off Phase 3 clinical trials for their Covid-19 vaccine Thursday following promising results from earlier trials indicating strong immune. A final analysis of Novavaxs UK Phase 3 trial results announced in March showed that the vaccine had an overall efficacy of 897 -- and its efficacy was 964 against the original coronavirus.
The Texas Vaccine Scheduler helps Texans get scheduled for a COVID-19 vaccine at clinics hosted by participating Texas public health entities. Register online at GetTheVaccinedshstexasgov. You will be notified by email or text when and where to get the vaccine.
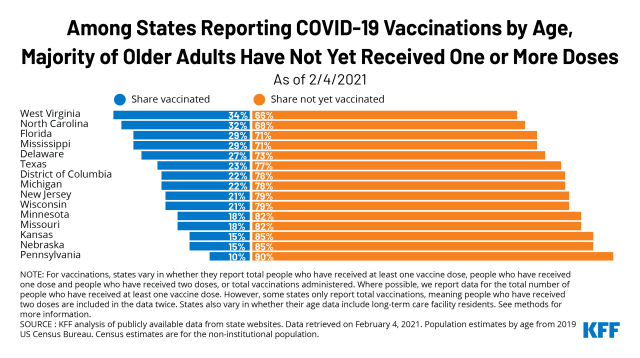
At This Early Stage Of The Covid 19 Vaccine Roll Out Most Older Adults Have Not Yet Been Vaccinated As Supply Remains Limited Kff

Covid Vaccines How Much They Cost Who S Bought Them And How They Re Stored

The Covid 19 Vaccination Line An Update On State Prioritization Plans Kff

Fifth Covid 19 Vaccine Reaches Phase 3 Development In Us

Covid 19 Vaccine Phases Groups In Texas

U S Advisory Group Lays Out Proposal On How To Prioritize Covid 19 Vaccine

Texas Will Receive 332 750 First Doses Of Covid 19 Vaccines This Week Dshs Says Kxan Austin

Covid 19 Vaccine Update Pa Department Of Health Expands Phase 1a Wellspan Health
State Coronavirus Vaccine Information Longview Tx

North Texas Woman Launches Crowdsourcing Site On Where To Get The Covid 19 Vaccine Kxan Austin
Latest U S Authorizes Booster Shot For People With Compromised Immune Systems

Posting Komentar untuk "What Phase Is Covid 19 Vaccine In Texas"